4 - Impact of keratin network regulation on migrating cells
Uniklinik RWTH Aachen (RWTH) - Germany
Student: Anne Pora
Supervisors: Rudolf Leube, Reinhard Windoffer, Björn Kampa, Bernd Hoffmann
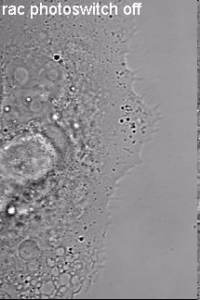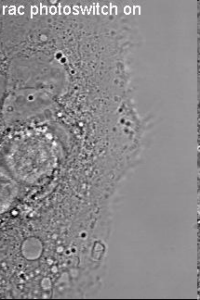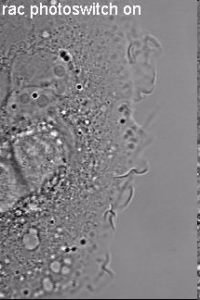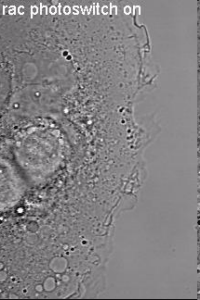
Induction of cell border dynamics through UV-light induced activation of pa-rac.
Cell migration is a highly complex process, in which physical and chemical signals act on different cellular components. We aim at understanding how the dynamic behavior of the epithelial keratin intermediate filament cytoskeleton and its associated hemidesmosomal anchorage sites is integrated in migrating cells and influenced by the mechanical characteristics of the environment.
Using human primary keratinocytes grown on extracellular matrices of different elasticities, we demonstrate that matrix stiffness affects cell morphology, speed and directionality of migration and keratin intermediate filament turnover. We find that hemidesmosomal components are organized in an ordered fashion in these cells exerting distinct forces on the matrix. We assess the effects of matrix stretching and of confinement on keratin network dynamics. Our data provide evidence for reorganization of the keratin cytoskeleton and its associated matrix adhesion sites in migrating cells responding to mechanophysical matrix properties.
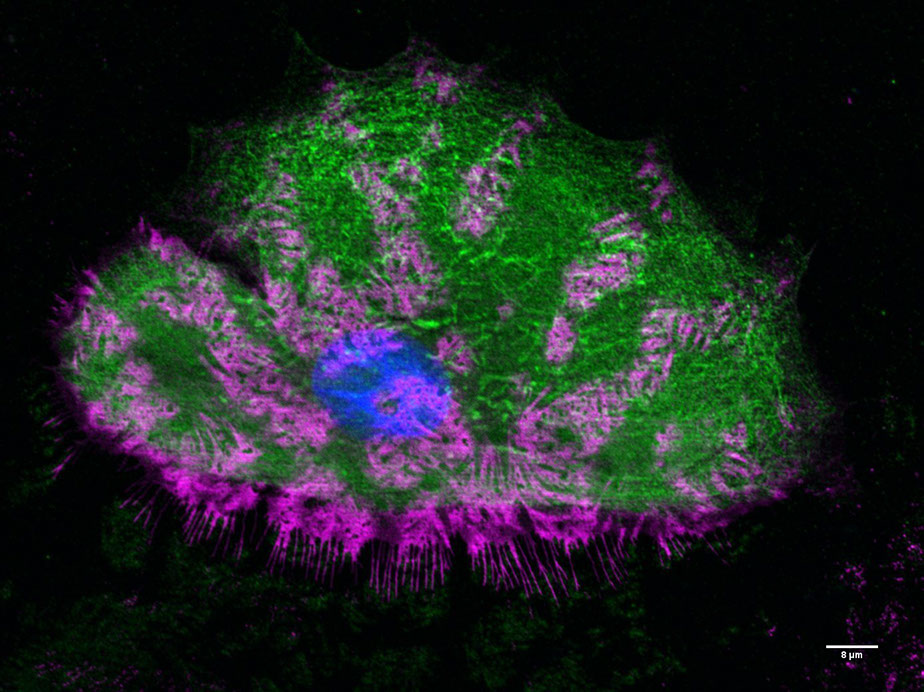
Fixed nHEK cell seeded on glass with fibronectin coating Immunostaining for Integrin-beta 4 (in magenta) and keratin (in green), and Hoechst staining (in blue)
Last update: 28.05.2018
incem@rwth-aachen.de
Advanced cell migration assays (P1)
Chemotaxis and 2D/3D Migration (P2)
Analysis of keratin dynamics during migration (P3)
Impact of keratin network regulation on migrating cells (P4)
Correlation analyses of migration structure components and front-rear interplay (P5)
Life cycle analysis of actin, focal adhesions and force measurements (P6)
Monitoring of cancer cell migration in living animals (P7)
Principles of the filopodia structure, dynamics and mechanics (P8)
Mechanisms of downstream signalling from the Rho GTPase network to
cell morphogenesis and cell motility (P9)
Real-time tracking of keratinocyte migration and analysis of cell membrane shape changes (P10)
Image analysis of integrated cytoskeletal network dynamics (P11)
Coupling bulk-surface models for cell migration (P12)
Shaping membranes and actin fibres by forces (P13)
Integrating shape change models and imaging – inverse problem solving and model validation (P14)
Understanding spatio-temporal dynamics of the cytosol network during cell migration (P15)
This project has received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Sklodowska-Curie grant agreement No 642866.